Issue 6: Why breaking into Regulatory Affairs is "that hard"?
Everyone says breaking into Regulatory Affairs is “hard.” And honestly? They’re not wrong.
Everyone says breaking into Regulatory Affairs is “hard.” And honestly? They’re not wrong. I keep hearing the same thing from people trying to get their foot in the door: companies just don’t hire entry-level. But there’s more to it than that. In this issue, we’ll explore why mentoring (or the lack of it) makes a difference, why piling on certificates won’t make you stand out, and what actually helps you think and sound like someone already in Regulatory Affairs. Plus, I’ll suggest a LinkedIn mentor whose voice is worth following if you’re starting out.
Today you will read in this issue:
a quote worth reflecting on
a recall worth learning from
a career advice worth exploring
a linkedin voice worth following
A quote worth reflecting on
Everyone says breaking into Regulatory Affairs is “hard.” They’re right, but probably not for the reason you think.
It’s not that you’re unqualified. It’s not that companies doubt your potential. The real barrier is often this: mentoring is hard work, and not everyone is willing to do it.
Teaching someone to think like an RA professional, explaining the reasoning behind decisions, walking through the nuances, takes patience and effort. Even experienced people can feel unsure of themselves when asked to guide a beginner. So instead of investing in that process, many companies default to “we need someone who can hit the ground running.”
That’s a failure of the industry, not a reflection of you.
So if you’re knocking on doors that won’t open: keep going. The right organization will have people willing to invest in you. And when you find them, you’ll grow faster than you imagined.
A recall worth learning from
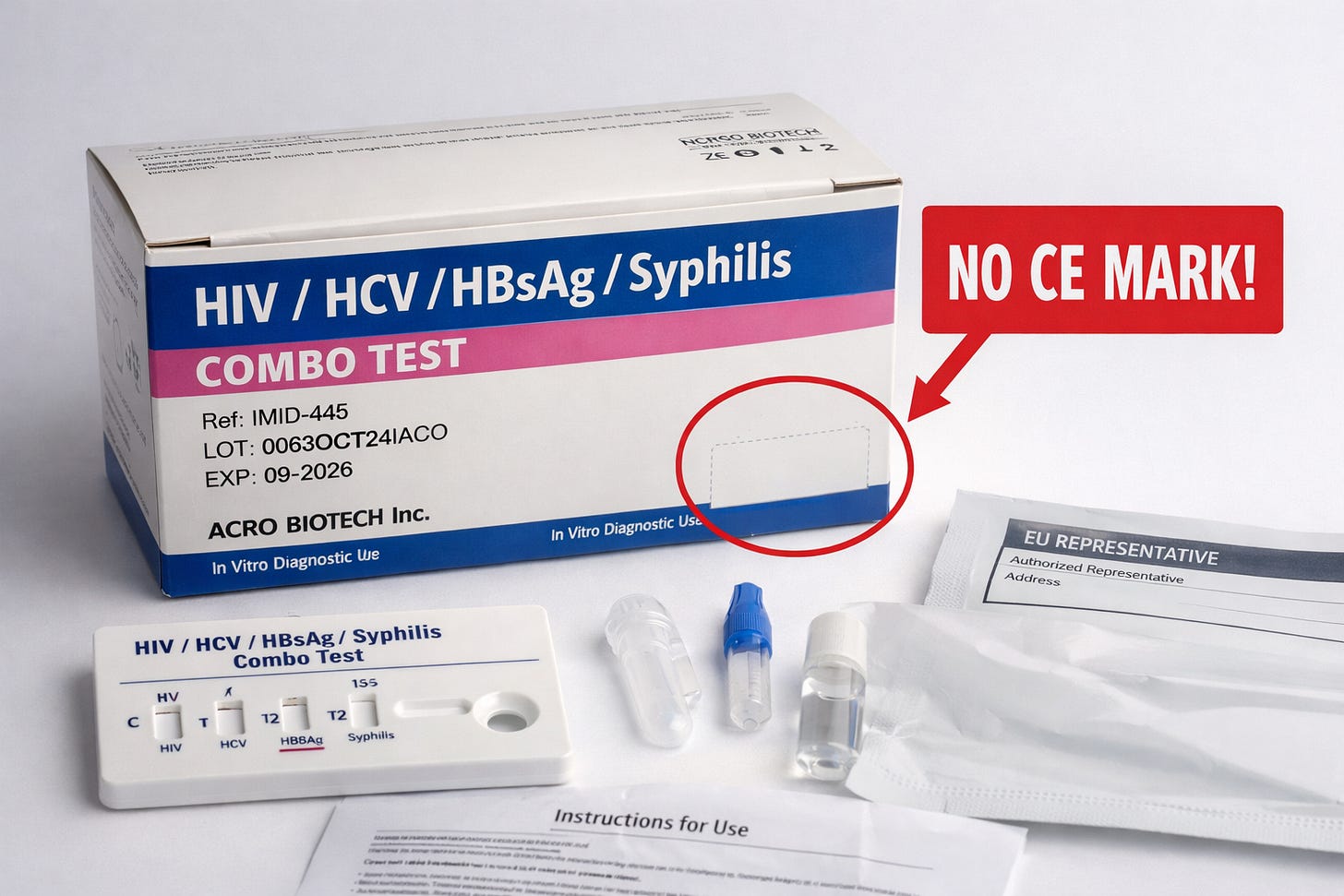
This could be an obvious mistake, but it happened.
Several rapid diagnostic tests in Greece including HIV, Syphilis, and LH self-tests were recalled because they were missing the CE mark and EU authorized representative info on their packaging and instructions.
The tests themselves work, but without proper labeling, traceability and regulatory compliance are broken. EOF, Greek authority, ordered the recall, and the distributor had to withdraw affected batches and keep records.
It’s a reminder that in regulatory affairs, even small oversights like a missing CE mark can trigger recalls and shake trust, even if the product functions perfectly.
Career advice worth exploring
Let me be honest: most people trying to enter Regulatory Affairs are doing the same things.
They take another course.
They add another certificate.
They wait until they “feel ready.”
And then they wonder why nothing happens.
Unfortunately, qualifications alone don’t make you stand out. Yes, you need them.
But, they make you look like everyone else. You need a differentiator based on your qualifications.
What actually gets attention is this: proof that you already think like someone in Regulatory Affairs.
Pick any product. A diagnostic test. A cosmetic. A supplement.
Now ask yourself:
How is this regulated in your country of interest?
What is the intended purpose?
What changes if that intended use shifts slightly?
If you can talk through these questions, you’re already ahead of most applicants.
A Linkedin voice worth following
Angelina Lisandrelli is a recognized expert in MedTech, not just because of her 15+ years in the field, but also because she’s a dedicated mentor. She regularly shares her insights through multiple newsletters, making complex topics approachable. I highly recommend following her if you want to stay informed and inspired.
Before you go, tell me:
That is it for this week! :)
Have a great week and see you in the next issue!


